|
OBJECTIVES:
1. define acids and bases using BOTH Arrhenius and Bronsted-Lowry definitions 2. predict products in acid-base reactions that form water 3. define pH and use the hydrogen or hydroxide ion concentrations to calculate the pH of a solution 4. distinguish between degrees of dissociation for strong and weak acids and bases 5. name simple acids and bases |
Definitions and NamingThrough time, definitions change. Especially ones that describe a set of actions. Visit the link and become familiar with Arrhenius and Brosnsted-Lowry acids. Going to college? Check out Lewis acids too.
Use your flow charts to be able to name acids and bases. HINT: if the formula starts with an H, its most likely an acid. If it ends with OH, its probably a base.
|
Predicting products of acid base reactionsLooking at the reactants you should be able to predict products from an acid and a base. Take the H+ and the OH- and put them together to make water. The pieces that are left? Look at the charges and form a compound. This of course changes the pH.
|
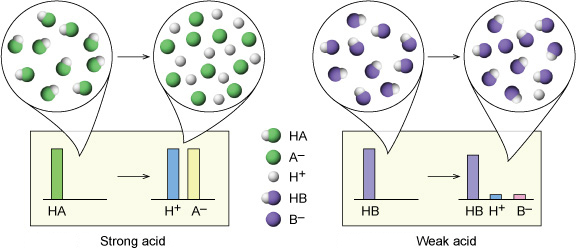
Strong VS Weak?
|