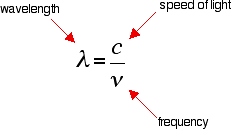Wavelength and FrequencyTake note of the differences in not only color, but wavelength and frequency.
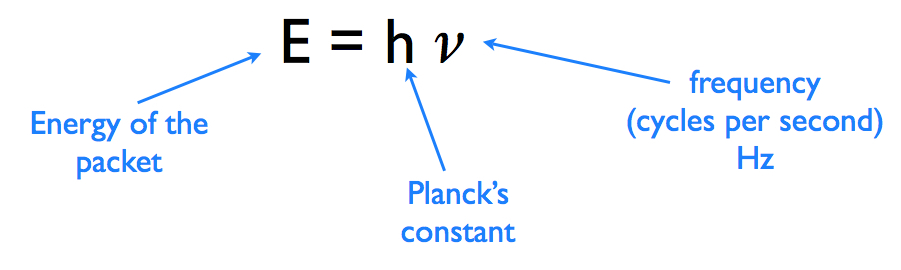
What happens when the wavelength gets shorter? What happens when the waves come more frequently? OBJECTIVES: 1. understand the electromagnetic spectrum and the mathematical relationships between energy, frequency, and wavelength of light 2. calculate the wavelength, frequency, and energy of light using Planck’s constant and the speed of light |
See Some Examples
|
As a distant star moves away from Earth, the light given off by the star has a measurably
lower frequency. What happens to the wavelength and energy of the photons of light when the frequency becomes lower? ANSWER: The wavelength becomes longer The energy decreases |
Which of these statements does not correctly describe a ray of light?
1. The energy of the light is directly related to its frequency. 2. As the wavelength of light increases, the frequency of light decreases. 3. Different colors of light carry different amounts of energy. 4. As the frequency of light increases, the speed of light decreases. ANSWER: The first three statements are true. Notice the final statement does not match up to the math equations above. |
A photon of light is determined to have a frequency of 6.24 x 10^14 Hz. What is the wavelength of this photon of light?
Notice that you could begin with either equation because you are given frequency. However, only one equation also has wavelength. Look up the speed of light (formula chart in the resources section) to solve the problem. ANSWER: 4.81 x 10-7 meters |