|
Energy is all around you, from the wind moving in the air to your own feelings of hot and cold. In fact the sensations of hot and cold are simply a detection of which way energy is flowing.
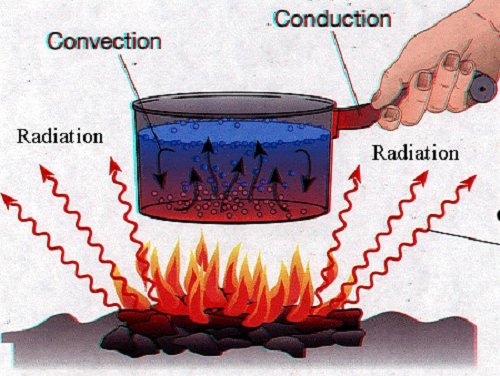
OBJECTIVES: 1. Identify different forms of energy such as heat, light, chemical, and kinetic. 2. Identify and explain the different processes of heat transfer (convection, conduction, radiation) 3. Determine if chemical reactions are exothermic or endothermic 4 Use the equation Q = m Cp (Tf - Ti) 5 use calorimetry to calculate the heat of a reaction |
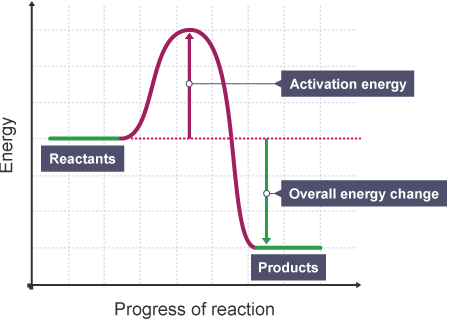
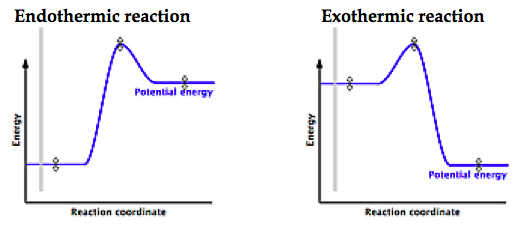
Endothermic VS Exothermic
Which way is the heat going? If its into the ice, you FEEL COLD because the ice is taking your heat to become water. Likewise, if it feels hot, the reaction is GIVING off energy and is exothermic.
|
Calculating HeatIn the video below... the calculating heat examples are taken to the experimental level. Rather than attempting to do this highly sensitive, somewhat dangerous, and relatively unexciting experiments on our own... we're going to youtube it. The guy doesn't breathe, but injects some excitement into something otherwise uninteresting and relates it to traveling to Mars.
|