|
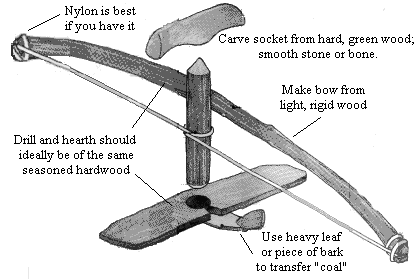
Using a bow and drill to make fire by friction.
|
Without fire, man would likely be conquered by the wild. Infectious diseases would run rampant, the terrain would prove unmanageable, and wild beasts would out muscle us. Fire allowed us to see in the dark, change the landscape, scare the beasts, and most importantly -- create new things. Simple clay was morphed into pottery, sand was turned to glass, ashes to soap, and from rocks we took metals.
Most of you know very little about fire other than it is hot. This section aims to change that and launch our chemical revolution. |
CREDIT FOR THIS SECTION
|
1. Journal credit.
-You have evidence of detailed notes -50 points -You have a summary of key points (paragraph form) 25 points -You have further questions that are not answered in the notes. 1 question- 5 points 2 questions 15 points 3 questions 25 points *these questions SHOULD NOT BE answerable by the notes *these questions SHOULD NOT BE common knowledge (or common sense) *who and where questions will not be accepted in any form (who invented fire / where was fire found) |
2. Fire Quiz
This will consist of 3 questions on any topic discussed above such as: classifying, chemical equations, the difference between oxidation and reduction, and the difference between charcoal and ash, and any linked vocabulary terms. You may retake quizzes once per week until they are passed or the grading period closes. ***You must pass this quiz to join a tribe for the project.*** Otherwise you may perform it alone. |
3. Fire Project
In order for chemists to exist, we must master fire. You may choose any of the below methods to create fire, and since you are essentially a Neanderthal in chemistry at this point, you may work as a Tribe. That is, up to 4 people may work together. You may practice outside of class, but it must be done IN CLASS to receive credit. 100: creation of actual flames 90: creation of a live ember 80: you made smoke 70: hot to the touch 50: you made a kit **ANY METHOD OTHER THAN FRICTION WILL NOT BE ACCEPTED** |
Fire basics: there are three things needed for fire: fuel, air, and heat. From a chemical perspective for the combustion of wood we have wood + air + heat --> ash + smoke + more heat
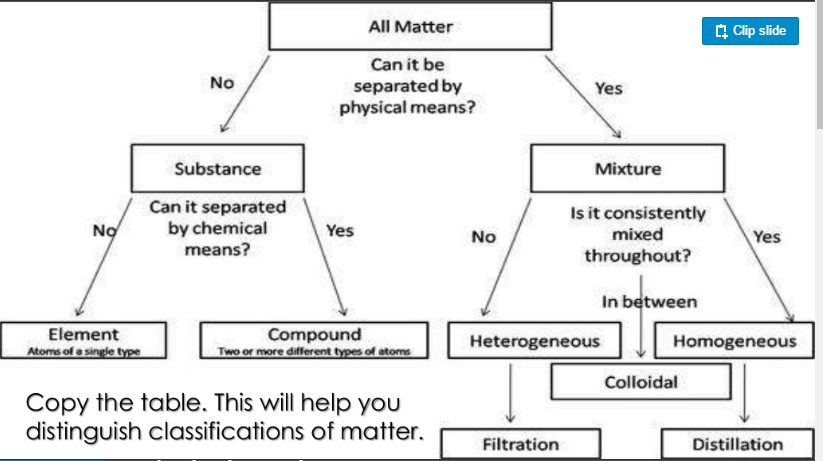
This seems fairly basic until we start looking at the process and what is happening. First off, there are very few pure substances in nature. If you pick up a piece of wood it is likely to have lichens, pollen, moss, dirt, etc on it. Therefore in order to fully understand what is happening and why differences occur we need to be able to classify materials according to their purity. Take notes on the pictures below to distinguish between these types of substances, the picture to the left suggests a method for "purifying" these mixtures.
This seems fairly basic until we start looking at the process and what is happening. First off, there are very few pure substances in nature. If you pick up a piece of wood it is likely to have lichens, pollen, moss, dirt, etc on it. Therefore in order to fully understand what is happening and why differences occur we need to be able to classify materials according to their purity. Take notes on the pictures below to distinguish between these types of substances, the picture to the left suggests a method for "purifying" these mixtures.
Nearly all things in nature are considered heterogeneous, they are NOT uniform in their composition. When you look at a piece of wood for your fire... the rings shown are an obvious difference of dark and light bands of color corresponding to the growth of the plant. This variation is what makes wood heterogeneous even if you were to clean off the dirt, pollen, etc. Even if we put one ring under a microscope you will find the different plant fibers and cells are not the same.
There are homogeneous natural substances, but they are few. Air is considered one of them because if a sample is taken, the parts of the air spread out evenly within the container. This is not to say that ALL AIR is the same, just that samples of it are (assuming the air is clean and that airplanes, bugs, and such excluded).
All things are possible to be purified, although this is difficult. For example, the air in your home is purified by filtration as it passes through the filters on your air conditioner. This is the reason that different levels of filter are sold, it depends on the level of purity the homeowner needs based on the SIZE of the particles that their allergies are sensitive to and conditions of their home. Purifying the wood is much more difficult, it needs to be ground into a pulp and then stirred until uniform (this is what the blender does when making a smoothie). Further, when put under a microscope we would still be able to pick out impurities. With that, test yourself on this idea:
There are homogeneous natural substances, but they are few. Air is considered one of them because if a sample is taken, the parts of the air spread out evenly within the container. This is not to say that ALL AIR is the same, just that samples of it are (assuming the air is clean and that airplanes, bugs, and such excluded).
All things are possible to be purified, although this is difficult. For example, the air in your home is purified by filtration as it passes through the filters on your air conditioner. This is the reason that different levels of filter are sold, it depends on the level of purity the homeowner needs based on the SIZE of the particles that their allergies are sensitive to and conditions of their home. Purifying the wood is much more difficult, it needs to be ground into a pulp and then stirred until uniform (this is what the blender does when making a smoothie). Further, when put under a microscope we would still be able to pick out impurities. With that, test yourself on this idea:
|
SELF QUIZ: classify each substance as heterogeneous or homogeneous.
1. Soil (a handful of dirt from outside) 2. Charcoal (the kind you buy for your grill) 3. Oceanwater (the kind fish swim in, not sonic drink) 4. Soda (Dr. Pepper, Pepsi, Sunkist, etc.) 5. Glass (windows, bottles) 6. Blood (the stuff vampires drink) 7. Bones (yes, the ones you need to stand) |
Check your answers. Be honest with yourself or it will be difficult to pass an exam.
1. heterogeneous 2. homogeneous 3. heterogeneous (homogenous only when filtered) 4. heterogeneous (you can see the bubbles) 5. homogeneous 6. homogeneous (unless under a microscope, then blood cells differ from blood plasma) 7. heterogeneous (bones have marrow in them) |
Solutions VS Pure Substances
Both solutions and pure substances appear the same to the eye. They are both uniform compositions as far as we can see with our eyes. The key difference is that a solution is a mixture that has been blended so well that we can't tell the difference. Take Kool-Aide for example, a solution of of water, sugar, and flavorings. The solution is uniform in any sample from the pitcher that is made, but is different from one pitcher to the next.
Compare that to pure substances and you will find that the composition is both uniform and fixed, it can't change from one pitcher or batch to the other. Take water, if filtered to its pure state it is always made of H2O. It doesn't matter who made it or where it came from, it is always the same. This is also the idea behind quality control for products, if one bottle of coke was different from the next bottle, people would stop buying it. This is actually a job that employs a chemist to ensure that all of the coke solutions are the same.
Separating solutions is not always as simple as filtration (separating by size). Some others we will explore are distillation (separating by boiling point) and re-crystallization (separating by solubility temperatures).
Both solutions and pure substances appear the same to the eye. They are both uniform compositions as far as we can see with our eyes. The key difference is that a solution is a mixture that has been blended so well that we can't tell the difference. Take Kool-Aide for example, a solution of of water, sugar, and flavorings. The solution is uniform in any sample from the pitcher that is made, but is different from one pitcher to the next.
Compare that to pure substances and you will find that the composition is both uniform and fixed, it can't change from one pitcher or batch to the other. Take water, if filtered to its pure state it is always made of H2O. It doesn't matter who made it or where it came from, it is always the same. This is also the idea behind quality control for products, if one bottle of coke was different from the next bottle, people would stop buying it. This is actually a job that employs a chemist to ensure that all of the coke solutions are the same.
Separating solutions is not always as simple as filtration (separating by size). Some others we will explore are distillation (separating by boiling point) and re-crystallization (separating by solubility temperatures).
|
SELF QUIZ: classify each substance as solution or pure substance
1. Charcoal 2. Oceanwater 3. Soda 4. Diamond 5. Blood |
Check your answers. Be honest with yourself or it will be difficult to pass an exam.
1. pure substance 2. solution (salt-water) 3. solution (sugar-water) 4. pure substance (assuming colorless) 5. solution (water |
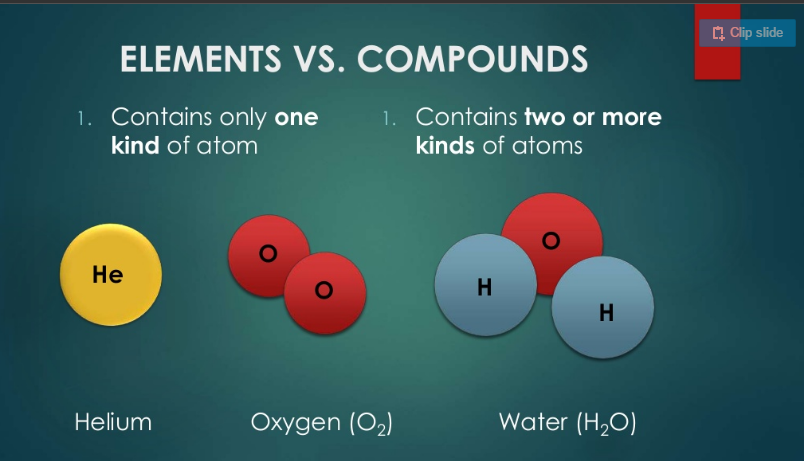
Compounds VS Elements
The difference between these two things is illustrated in the picture at the right. This is often the most mis-understood concept from middle school. Simply put, a compound has more than one element inside of it whereas an element has only one type of element and is listed on the periodic table. of Examples:
Diamond: C (element) water: H2O (compound) quartz: SiO2 (compound)
Oxygen: O2 (element) celluslose: CH2O (compound) carbon dioxide: CO2 (compound)
Where one element stops and another begins can be distinguished in its abbreviation by the number of capital letters in its formula.
The little subscripts in the formula tell you how many of each element make up the fixed ratio. For water that is two hydrogen atoms for every one oxygen atom.
The difference between these two things is illustrated in the picture at the right. This is often the most mis-understood concept from middle school. Simply put, a compound has more than one element inside of it whereas an element has only one type of element and is listed on the periodic table. of Examples:
Diamond: C (element) water: H2O (compound) quartz: SiO2 (compound)
Oxygen: O2 (element) celluslose: CH2O (compound) carbon dioxide: CO2 (compound)
Where one element stops and another begins can be distinguished in its abbreviation by the number of capital letters in its formula.
The little subscripts in the formula tell you how many of each element make up the fixed ratio. For water that is two hydrogen atoms for every one oxygen atom.
CHEMISTRY OF FIRE
Back to our burning wood. We can now express this reaction in chemical terms instead of generic words like wood. Since most of this wood is cellulose, CH2O, and the air is a solution of (mostly) nitrogen and oxygen, O2. During the combustion (burning) the nitrogen in the air is the same going out of the fire as it is coming into the fire we ignore it. So we can now write a chemical equation for the substances changed in the reaction:
FIRST reaction: CH2O(s) + O2(g) --> CO2 (g) + H2O(g)
This is read as "1 part cellulose (solid) burns in 1 part oxygen (gas) to produce 1 part carbon dioxide (gas) and 1 part water (gas) as products. The (s) and (g) tell us what phase the substance is in (s) solid (g) gas (l) liquid.
What about ash and smoke, aren't they produced too? Well no, not if the cellulose is completely combusted. Remember however that the wood is MOSTLY cellulose, the other nutrients and minerals in the wood that aren't burned. Also, if there isn't enough oxygen getting to the cellulose then tiny pieces of charcoal are made in the form of smoke. This is why if you blow into a fire it reduces the amount of smoke and burns more brightly. So if we just heat the wood without oxygen at all, it doesn't burn, but it produces charcoal.
SECOND reaction: CH2O(s) -----> C(s) + H2O(g)
Here we produce charcoal instead of carbon dioxide. This is not the same as ashes, which are minerals that do not burn. Charcoal is the same stuff that CAN BE BURNED and you buy it for your cookout.
THIRD reaction: C(s) + O2(g) -----> CO2(g)
If you look closely, the first reaction is the sum of reactions two and three. So we can get two different products by manipulating the conditions. The first is OXIDIZING (we add oxygen to the reaction), the second is REDUCING (we do not use oxygen). A fire that is oxidizing (has lots of air in this case) will burn and leave ashes behind which are white or gray non-flammable and a mixture of different minerals. A fire that is reducing (heated without oxygen) produces charcoal which is black, flammable, and mostly carbon. A fire with a little bit of air produces a little bit of both, which can be separated or re-burned. An easy way to separate them is to take advantage of differences in their other properties, ashes either dissolve in water or sink to the bottom. Charcoal has air pockets inside that make it less dense than water so it floats. These properties can be exploited in one of our future project options: potash.
Fire PloughThe most basic of fire making techniques. Also one of the hardest to perform.
|
Bow and Drill
|
PreservationOnce you have fire... keeping it is better than restarting it. This was an ancient job assigned to women "keep the home fires burning." Later this was used for match-lock muskets. Often referred to as a slow match.
**EXTRA CREDIT ONLY** You may earn a 15 point bonus certificate by completing this project. Research and create a slow match. |