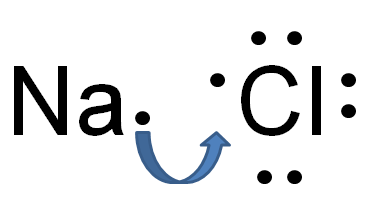Objectives:
1. Write formulas for ionic compounds
2. Write formulas for compounds
3. Write formulas for polyatomic compounds
1. Write formulas for ionic compounds
2. Write formulas for compounds
3. Write formulas for polyatomic compounds
REMEMBER THE FIRST STEP IS ALWAYS IDENTIFY THE TYPE OF COMPOUND: ionic, covalent, or polyatomic?
All formulas come from stable Lewis dot structures.
Ionic FormulasMetals are always written first, they are less electronegative.
If the charges are equal, you get one of each element such as Na+ and Cl- yields NaCl. Unequal charges yield a criss-cross effect. The charge of one becomes the amount of the other. EX: Mg2+ and Br- yields MgBr2 Transition metals will have Roman Numerals in their name to give you the charge. EX: Iron III Oxide Ions: Fe3+, O2- Formula: Fe2O3 |
Covalent FormulasCovalent formulas come from their names and greek prefixes since there is more than one possibility through sharing.
EX: CO = Carbon monoxide CO2 = Carbon dioxide NO = nitrogen monoxide N2O2 = dinitrogen dioxide 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6= hexa 7 = hepta 8 = octa 9 = nona 10 = deca |
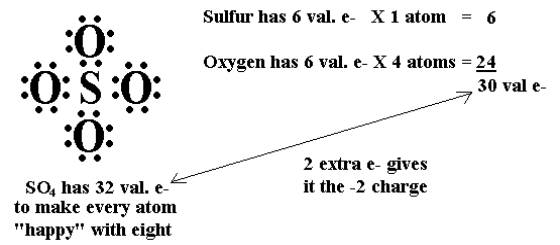
Recognizing common polyatomic ionsThese occur when a covalent combination requires extra electrons in order to be stable. In the above image, SO4 requires 2 extra electrons so it gets a 2- charge.
Since they have a charge, these molecules ionicly bond with a metal EX: Iron II Sulfate = FeSO4 EX: Iron III Sulfate = Fe2(SO4)3 You should recognize the common ones listed on the back of your periodic table. The most common we will encounter in class and should be MEMORIZED: OH- = hydroxide SO42- = sulfate NO3- = nitrate |