|
Objectives:
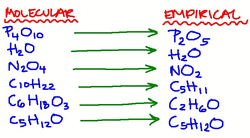
1. Calculate the % by mass for any element in a compound. 2. Calculate the molecular formula for a compound 3. Calculate the empirical formula for a compound Definitions: Molecular: a formula giving the number of atoms of each of the elements present in one molecule Empirical formula: a formula giving the RATIOS of the elements present in a compound |
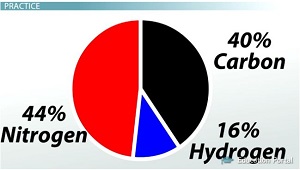
Calculating percentageCalculated in the same way you have been doing your grades all year. Take the portion you want, divide by the total, then multiply by 100.
|
Empirical FormulaDetermining the ratios comes from the percentages. If you divide all by the smallest percentage present, then you have the ratios. IT MUST BE DONE IN MOLES!
|
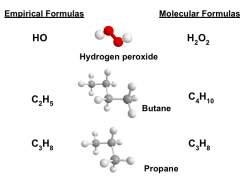
Molecular FormulaTo get the MOLECULAR formula, you must know the EMPIRICAL formula.
Then divide their masses to get the ratio. Multiply the empirical formula by this ratio. |