|
Home-made soap rendered from animal fat and wood ash. Courtesy of: Practical Primitive
|
Primitive soaps weren't actually used for hand washing and body cleansing as we know them now. Mostly because primitive societies (including the Romans and Greeks here) didn't quite understand the effects of bacteria and disease, and most commoners didn't even know of bacteria's existence. Instead of soap they relied on urine, plants, clays, potash, and soda for doing things like laundry. Yes that's right, urine... also used as a teeth whitener by the Romans.
So why soap? To clean the sheep wool used for clothes of its grease and dingle-berries that interferes with the dyes used to make different colors -- colors that indicated you were wealthy and of high status. |
CREDIT FOR THIS SECTION
|
1. Journal credit.
-You have evidence of detailed notes -50 points -You have a summary of key points AND the MSDS for Lye (sodium hydroxide CAS 01310-73-2) with dangers to eye contact, skin contact, inhalation, and ingenstion 25 points -You have further questions that are not answered in the notes. They must also not be deemed as common knowledge or common sense questions. 1 question- 5 points, 2 questions 15 points, 3 questions 25 points |
2. QUIZ
This will consist of 3 questions on any topic discussed below such as: polarity, solubility, how to balance an equation, any linked vocabulary terms You may retake quizzes once per week until they are passed or the grading period closes. ***You must pass this quiz to perform the project.*** |
3. SOAP PROJECT
You have the choice of making either black soap or white soap. Should you choose to make white soap, I will happily provide you with the Sodium Hydroxide and lab equipment. Should you choose the black soap and use potash for your creation, you will receive a 15 point bonus certificate. |
Soap Basics
We generally need our potash (potassium carbonate) or washing soda (sodium carbonate) as our precursor to soap creation, and some of you have tasted and felt its similar qualities, but it doesn't clean very well. However, our cave-geniuses watched our cave-dunces playing in the dirty fire pit and saw that when they were cooking animals, fat dripped down into the potash and this is what really got them clean. An interesting anecdote is that soap made from potash creates a black liquid soap, while soap from soda creates a white solid.
Soap was also extremely important to American History as westward expansion opened new areas for agriculture that fed the rapidly growing industrial towns. Anywhere cloth was being made from wool, there was a demand for soap. Anywhere the massive cotton boom fueled by expansion and Eli Whitney's cotton gin provided cotton for cloth, there was a demand for alkali's to help the dye process that gave color. Additionally two of the oldest (and you'll probably recognize their names) chemical companies in the world started out in the American soap and candle making business. They were started by William Colgate (of toothpaste fame) in 1806 in New York, and William Proctor and James Gamble in Cincinnati (of Tide and P&G fame).
Soap was also extremely important to American History as westward expansion opened new areas for agriculture that fed the rapidly growing industrial towns. Anywhere cloth was being made from wool, there was a demand for soap. Anywhere the massive cotton boom fueled by expansion and Eli Whitney's cotton gin provided cotton for cloth, there was a demand for alkali's to help the dye process that gave color. Additionally two of the oldest (and you'll probably recognize their names) chemical companies in the world started out in the American soap and candle making business. They were started by William Colgate (of toothpaste fame) in 1806 in New York, and William Proctor and James Gamble in Cincinnati (of Tide and P&G fame).
We have already mentioned bases, and a concentrated form of this is chemically known as Sodium Hydroxide with formula NaOH. This is one the most important and widely used chemicals today and might be found under its ancient common names lye and caustic soda. This stuff is so reactive and corrosive, it is also used as a drain cleaner.
Chemistry of Soap
We mentioned solubility in POTASH, but didn't talk about why some things were soluble and some things weren't. In general we can classify compounds as ionic, polar, and non-polar. The idea of polarity is key to understanding this idea, which basically states that the electrons shared in the bond between the atoms is unevenly shared, creating charged regions of the compound. Water has this property, and in doing so has become the essence of life and gives it some of its unique abilities such as adhesion, where it will stick to itself because the positive end of one water molecule is highly attracted to the negative end of the next water molecule.
|
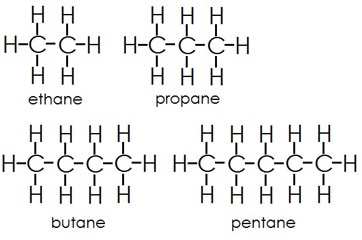
A non-polar example would be any oil, made up primarily of long carbon chains with hydrogen atoms attached all along the chain. Since hydrogen and carbon have almost the same electronegativity they share the electrons nearly evenly and no charged regions are created. Again, if you plan on moving to AP Biology, you had better master these ideas.
|
Back to solubility... compounds that are "like" each other in terms of polarity (either polar or non-polar) tend to dissolve in each other. So ionic and polar substances tend to dissolve in each other (like salt in water), and non-polar substances tend to to dissolve in each other (like oil in gasoline).
So water's polarity becomes extremely important to us not just for performing chemical reactions, but also for life itself. If our blood wasn't primarily water, it wouldn't be able to deliver the appropriate nutrients to our cells as they wouldn't dissolve. Further those same cells you were studying in biology... the membrane is made up of non-soluble fats and oils (so they don't dissolve away...). (Thinking of AP Biology? Go ahead and look up lipids and triglycerides now)
That brings us back to soap... we have to find a way to bond with (attach to) BOTH polar and non-polar substances. Good soaps are such long polymer chains that the middle is effectively neutral, one end is polar, and the other end is non-polar. This is a great simplification, but the specifics can be found under saponification if you are interested.
So water's polarity becomes extremely important to us not just for performing chemical reactions, but also for life itself. If our blood wasn't primarily water, it wouldn't be able to deliver the appropriate nutrients to our cells as they wouldn't dissolve. Further those same cells you were studying in biology... the membrane is made up of non-soluble fats and oils (so they don't dissolve away...). (Thinking of AP Biology? Go ahead and look up lipids and triglycerides now)
That brings us back to soap... we have to find a way to bond with (attach to) BOTH polar and non-polar substances. Good soaps are such long polymer chains that the middle is effectively neutral, one end is polar, and the other end is non-polar. This is a great simplification, but the specifics can be found under saponification if you are interested.
To make your soap... you need fat(s) + 3 lye (aq) ---> 3 sodium-fats(aq) + glycerol(aq) which was likely discovered after cooking meat over the fire and the fats dripped down into the coals. You can now go read your soap/shampoo bottle and find ingredient names starting with SODIUM XXXXXameate, where the XXXXX tells you which fat was used to make the soap.
|
We are starting to get a variety of coefficients in the chemical equations, for our soap there are 3s running about in front of both the lye and the sodium-fats. This is because the total amount of atoms that go in, also come out and from your FARTS acronym that rearrangement/reaction is referenced here. A variety of experiments can easily prove this by using a scale and capturing all the parts of a reaction both before and after and demonstrating that the masses are equal. The coefficients ensure that the count for each atom present is the same on both sides of the arrow, both the reagents (in) and products(out).
|
Tyler Dewitt makes balancing simple... notice how the color coded atoms rearrange to make a "reaction."
|
Practice Problems: balance the following equations so that the atoms you put in are the same atoms that you get out.
1. Fe(s) + Cl2(g) --> FeCl3(g)
2. H2 (g) + Cl2(g) --> HCl(g)
3. Fe(s) + O2(g) --> Fe2O3(s)
4. NaCl(aq) + Al(s) --> AlCl3(s) + Na(aq)
ANSWERS (Coefficients only)
1. 2, 3, 2
2. 1, 1, 2
3. 4, 3, 2
4. 3, 1, 1, 3
2. H2 (g) + Cl2(g) --> HCl(g)
3. Fe(s) + O2(g) --> Fe2O3(s)
4. NaCl(aq) + Al(s) --> AlCl3(s) + Na(aq)
ANSWERS (Coefficients only)
1. 2, 3, 2
2. 1, 1, 2
3. 4, 3, 2
4. 3, 1, 1, 3