|
A man spins fibers using a foot powered machine to do the twisting that give their strength.
|
It is believed that as long as 20,000 years ago clothing was made of spun fiber and not just animal skins. It is also believed that the first style of clothing was a simple skirt--consisting of a string belt with additional strings hanging from the waist to just above the knees. It was also useful for creating fishing line, setting animal traps, necklaces, and to fasten stones to sticks.
Twine, has its own unique set of properties and models some basically chemistry that we wouldn't otherwise be able to see quite nicely. Further, twine is the precursor to textiles which was one of the original motivators for the chemical industry. Early on it was the creation of dyes and colorfast clothes to distinguish the nobles and kings from merchants, peasants, and slaves. Today chemistry is creating synthetic polymers for superior and desired properties such as moisture wicking and durability. Later in the course, you will have the opportunity to make both your own dye and your own synthetic fiber. |
The Chemistry of Fibers
|
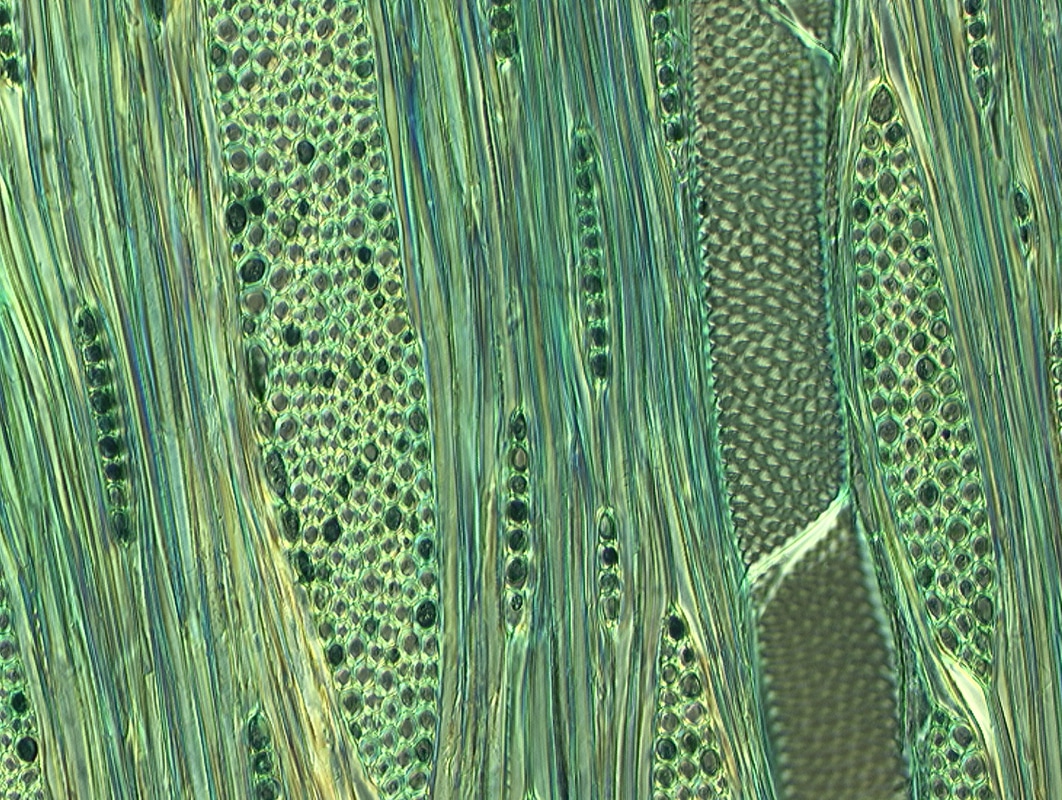
Prior to your modern synthetic clothes of Rayon, Nylon, etc. there were two major sources of fibers for ropes and clothing: those from plants, and those from animals. Cellulose (talked about during the FIRE component of this class as CH2O) we are familiar with, but an examination of its internal structure is warranted. Cellulose is made up of long chains of Carbon atoms with oxygen and hydrogen atoms attached to it as seen under a microscope here
|
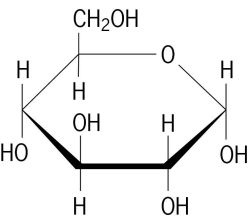
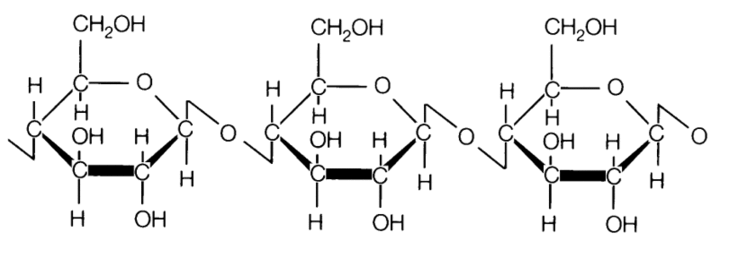
Cellulose cells can't be reused by a plant as they are. So in order for a plant to grow or repair itself it must convert other forms of matter into cellulose. In order to do this they can only use relatively small molecules like carbon dioxide, water, and soluble minerals that have been dissolved in water (the reason we use fertilizer to help them grow). You should recall photosynthesis from biology as an example of this process, where 6CO2 + 6H2O + light energy = C6H12O6 + 6O2. Recall that the coefficients in front of each compound are amounts of the whole molecule whereas the numbers below each letter are amounts of that particular element in ONE molecule. You should be able to count how many of each atom are present in the equation. So here we take a lot of things to make 1 sugar molecule and return 6 oxygen molecules (composed of 12 atoms) to the atmosphere. What the plant does, is use another chemical reaction called polymerization to change this glucose into cellulose by linking many glucose molecules together. Take a look at the many H and OH parts in the structure as this is where the link is made. One H from a glucose is paired with an OH from a second glucose to make water, and a bond is formed where the water was removed. Take a look at the pictures.
After linking many of these glucose molecules together, we get cellulose. This also helps us distinguish between the two forms and gives us a new way to classify when referring to chemicals: molecular and empirical formulas. The molecular formula of the larger structure is C6H12O6. It tells us how many atoms of each kind are present in the molecule. If you reduce this by noticing that there are double the amount of Hydrogens as there are Carbons and Oxygens you get the empirical formula for celluslose CH2O. This helps us to understand the biology better because big molecules are most easily built from smaller ones, and if we know what the smaller parts are we can devise ways to make bigger ones. So we can use small monomers to create repeating chains of polymers. When this happens, the true formula for cellulose appears at C6H12O6 which is essentially 6 of the original parts linked together. The key difference between the two formulas is that molecular formulas are the true amounts of each atom present and the empirical formula is the ratio of those elements.
This takes us to the other source: animals. Animals are made up of those amino acids (proteins) that you have likely heard about. Both wool and silk are made in the same way we just substitute proteins for the sugars that the plants use. A key difference here is that the proteins can be polymerized in any order to give different structures and properties. Such combinations include not only the fibers we are interested in for making fabrics and ropes, but also hemoglobin (used in your blood to transport oxygen), skin, and hair.
If you really liked biology, you will need this and other chemical ideas to be able to move on to AP Biology.
This takes us to the other source: animals. Animals are made up of those amino acids (proteins) that you have likely heard about. Both wool and silk are made in the same way we just substitute proteins for the sugars that the plants use. A key difference here is that the proteins can be polymerized in any order to give different structures and properties. Such combinations include not only the fibers we are interested in for making fabrics and ropes, but also hemoglobin (used in your blood to transport oxygen), skin, and hair.
If you really liked biology, you will need this and other chemical ideas to be able to move on to AP Biology.
An important distinction here is that animal HAIR is not the same as animal WOOL. They are composed of a different sequence of amino acids in their polymer and this gives them different properties. Wool is a fiber that clings to itself, hair tends to be smooth and slick and is designed for shedding rain. While it might get tangled, this is not the same as clinging (think hair on static filled clothes).
|
Self quiz
1. How many atoms of hydrogen are present in 6H2O? 2. How many atoms of oxygen are present in 6H2O? 3. How many atoms of oxygen are present in 6CO2 + 6H2O + light energy --> C6H12O6 + 6O2 |
Answers
1. There are 12 atoms of H. H = 6molecules x 2atoms/molecule = 12 atoms of H 2. There are 6 atoms of O. O = 6molecules x 1atom/molecule = 6 atoms of O. 3. There are 18 atoms of O. First, recall from middle school that the arrow sign splits the chemical reaction into before and after situations. Before the arrow atoms are arranged one way, and after the arrow they are arranged another way, but the number and kind of atoms remain the same. So there are 6 molecules of CO2, giving 6 x 2 = 12 oxygens in the carbon dioxide. There are also 6 oxygen atoms present in the water 6H2O. When we add the 12 oxygens from carbon dioxide to the 6 that are in the water molecules we have 18 total oxygen atoms. This equals the 6 oxygen atoms present in the glucose plus the 12 in elemental oxygen that are produced for us to breathe... for the same total of 18. |
General Chemical Safety
While there isn't really anything here that warrants a chemical safety hazard, (and so far your dangers have included not using common sense...) it is a good time to begin looking at how to determine if a particular chemical is safe. To do this, we review the material safety data sheet known as the MSDS. By law, all chemical manufacturers must create this for any hazardous chemical they either produce themselves or import from elsewhere.
As a general rule for the internet, anybody can post their own brand or interpretation of the truth, so ensure that when finding MSDS you are using reputable companies such as Flinn Scientific and Sigma-Aldrich. When searching online to ensure you are looking up the right chemical it is also beneficial to know the Chemical Abstracts number (CAS number) as this is used like a UPC bar code is at wal-mart, but for chemicals. I will do my best to remember to post such numbers throughout this course or a direct link to an MSDS. If I fail, I would appreciate the request... and may perhaps offer bonus points to those who find not only the failure, but also the proper CAS number.
MSDS will generally provide you way more information than you care to know, however the safety hazards summary is generally found in the third section. For your notes, please summarize the hazards for: charcoal (cas 7440-44-0), cellulose CAS: 9004-34-6, and potassium carbonate CAS: 584-08-7. You should include the identity of the company you got the msds from and the potential heath effects for eye contact, skin contact, inhalation, and ingestion.
As a general rule for the internet, anybody can post their own brand or interpretation of the truth, so ensure that when finding MSDS you are using reputable companies such as Flinn Scientific and Sigma-Aldrich. When searching online to ensure you are looking up the right chemical it is also beneficial to know the Chemical Abstracts number (CAS number) as this is used like a UPC bar code is at wal-mart, but for chemicals. I will do my best to remember to post such numbers throughout this course or a direct link to an MSDS. If I fail, I would appreciate the request... and may perhaps offer bonus points to those who find not only the failure, but also the proper CAS number.
MSDS will generally provide you way more information than you care to know, however the safety hazards summary is generally found in the third section. For your notes, please summarize the hazards for: charcoal (cas 7440-44-0), cellulose CAS: 9004-34-6, and potassium carbonate CAS: 584-08-7. You should include the identity of the company you got the msds from and the potential heath effects for eye contact, skin contact, inhalation, and ingestion.
Identifying chemical changes
It might also be useful to know if you have a chemical reaction going on that may warn you to be extra cautious, especially if you don't know what the products are. The acronym FARTS will be useful to you.
F: fizzing, if there is bubbling or fizzing happening, this is usually an indicator of chemical reaction. It is important to realize this is sudden and due to putting two things together and is different from intentional addition of heat (boiling---liquid to gas) or things coming out of solution (like gas bubbling out of your soda when you open it).
A: aroma, if there is a new smell that was different from before, chances are you have a chemical reaction. This is not the same as opening a lid and letting the gas particles (that you smell) expand.
R: rearrangement/reaction, if two atoms are switching places it is a clear indicator that the change is chemical. This is your best friend on paper tests, if you have products (after the arrow) that have atoms in different places than reactants (left side of the arrow) its an easy identifier. For example in the fire equations the oxygen is alone at the beginning and joins with carbon at the end so it has switched places.
T: Temperature change, again this must be sudden and not intentional. If you put it in the oven, on the stove, or over a fire then it doesn't count. If you put something together and it seems like 'magic' that it got hot or cold, then you have a chemical reaction. Giving off both heat and light count here.
S: Something new, this is the gold standard for a chemical reaction in reality. If two things come together and you get evidence of something new (like ash from a fire) then you definitely have a chemical reaction.
F: fizzing, if there is bubbling or fizzing happening, this is usually an indicator of chemical reaction. It is important to realize this is sudden and due to putting two things together and is different from intentional addition of heat (boiling---liquid to gas) or things coming out of solution (like gas bubbling out of your soda when you open it).
A: aroma, if there is a new smell that was different from before, chances are you have a chemical reaction. This is not the same as opening a lid and letting the gas particles (that you smell) expand.
R: rearrangement/reaction, if two atoms are switching places it is a clear indicator that the change is chemical. This is your best friend on paper tests, if you have products (after the arrow) that have atoms in different places than reactants (left side of the arrow) its an easy identifier. For example in the fire equations the oxygen is alone at the beginning and joins with carbon at the end so it has switched places.
T: Temperature change, again this must be sudden and not intentional. If you put it in the oven, on the stove, or over a fire then it doesn't count. If you put something together and it seems like 'magic' that it got hot or cold, then you have a chemical reaction. Giving off both heat and light count here.
S: Something new, this is the gold standard for a chemical reaction in reality. If two things come together and you get evidence of something new (like ash from a fire) then you definitely have a chemical reaction.
CREDIT FOR THIS SECTION
|
1. Journal credit.
-You have evidence of detailed notes -50 points -You have a summary of key points (paragraph form) 25 points -You have further questions that are not answered in the notes. 1 question- 5 points 2 questions 15 points 3 questions 25 points *these questions SHOULD NOT BE answerable by the notes *these questions SHOULD NOT BE common knowledge (or common sense) |
2. Twine Quiz
This will consist of 3 questions on any topic discussed above such as: what are plant and animal fibers composed of, difference between a molecular and empirical formula, how fibers can be spun, the difference between a monomer and a polymer, general safety, MSDS, indicators of a chemical reaction. You may retake quizzes once per week until they are passed or the grading period closes. ***You must pass this quiz to perform the twine project.*** |
3. Twine Project In caveman society this would be delegated out to an individual. While you may gather your materials together, the spinning of yarn would be done in a partnership. If you chose to use a drop spindle, this will be individual credit. (2 people for the project, 1 if you want the bonus certificate for the drop spindle) *purchase of commercial yarns and cords is forbidden in this project* you may however, use source material such as cotton or wool. You are also free to get creative with your material. Creation of either yarn or cord is acceptable for this project. Grade Yarn Cord or 2-ply yarn 100: 10 feet 5 feet 90: 9ft 4.5ft 80: 8ft 4ft 70: 7ft 3.5ft 50: 4ft 2ft **Quality control** If you submit your twine and it comes apart when I pull on it, you fail. The fibers MUST be interTWINEd. |
|
Many plant fibers work well such as: milkweed and nettles both of which grow in the many parks and open lands around Mesquite. Other plant fiber sources are the insides of dead tree bark (living don't work well...needs to be a dead tree)
15 bonus points are available for the project using naturally occurring plant fibers. You must submit the location, type of plant, and a photo of you harvesting the plant to claim this bonus. |
If you have a dog, you likely have a source of wool as well. This can generally be found in the chest and armpit region of the dog and is different than the hair on the back or head.
If you know a butcher, animal sinew is very good for this use and many ancients and natives used this for stringing their bows and making fishing line while saving the plants for less intense uses. 8 bonus points are available for using naturally occurring animal fibers. You must submit the type of animal, and the location on the body the fiber was harvested as well as a photo to claim this bonus. |
There are many good "how to" videos on youtube for spinning and a creating drop spindles. Should you create your own drop spindle, bonus certificates are available:
15 points from naturally occurring materials. 5 points from recycling modern materials. 0 points will be awarded for parts purchase and assembly. |
SIMPLE HAND SPUN PROCESS FROM WOOL
The simplest of fibers for spinning is using your pet's wool, or your friend's pet, or your uncle's pet... etc. The longer the hair and the larger your pet, the easier the harvest. If you want the lazy way out, go buy a bag of cotton fiber (not cotton balls, this is sometimes called batting and is found in the arts and craft sections of most stores like wal-mart and target).
To get the basic idea, take a handful of wool or cotton and pinch off a small amount and pull it until it separates from the rest of the collection. Take a close look at their length and how well they cling to each other as this is important to your own personal adaptation of this process. Set this to the side.
Pinch and pull another length, but do not allow it to separate from the main collection. Now twist it up and then grip it between your fingers and pull more out of the main collection. If you are doing this correctly, the twist should begin to go into the next batch of fiber that you would be pulling out from the main section. Alternating twisting and pulling is how you spin yarn and twine.
Once you reach about a foot, you need a way to hold and twist the yarn while still holding the collection of fiber and pulling it out. This is where making your own drop spindle comes into play, but you can get creative too, such as using a sibling or another cave-chemist to aide you. If I get around to it, I will make some that you can check out with a deposit.
If at any point you allow your twine/yarn to untwist, its strength and come apart. Thus you need to devise a way to keep it together: you can weave it for clothes/textiles or ply it for cord. To ply your twine (make another layer) you can spin one length clockwise (or righty-tighty) and one counterclockwise (lefty-loosey) then spin the two together into a single cord. This is the reason the grading scheme is the same credit for half the length of cord as plain yarn. If you want to preserve your yarn for textiles later but don't want it to unravel in the mean-time, you can twist the yarn A LOT, then fold it in half and let it twist back onto itself. A word of caution... you may want to practice this with a short piece first, because if you mess up you could lose your work.
As part of an exam, you may be asked how plants and animals create polymers from monomers.
To get the basic idea, take a handful of wool or cotton and pinch off a small amount and pull it until it separates from the rest of the collection. Take a close look at their length and how well they cling to each other as this is important to your own personal adaptation of this process. Set this to the side.
Pinch and pull another length, but do not allow it to separate from the main collection. Now twist it up and then grip it between your fingers and pull more out of the main collection. If you are doing this correctly, the twist should begin to go into the next batch of fiber that you would be pulling out from the main section. Alternating twisting and pulling is how you spin yarn and twine.
Once you reach about a foot, you need a way to hold and twist the yarn while still holding the collection of fiber and pulling it out. This is where making your own drop spindle comes into play, but you can get creative too, such as using a sibling or another cave-chemist to aide you. If I get around to it, I will make some that you can check out with a deposit.
If at any point you allow your twine/yarn to untwist, its strength and come apart. Thus you need to devise a way to keep it together: you can weave it for clothes/textiles or ply it for cord. To ply your twine (make another layer) you can spin one length clockwise (or righty-tighty) and one counterclockwise (lefty-loosey) then spin the two together into a single cord. This is the reason the grading scheme is the same credit for half the length of cord as plain yarn. If you want to preserve your yarn for textiles later but don't want it to unravel in the mean-time, you can twist the yarn A LOT, then fold it in half and let it twist back onto itself. A word of caution... you may want to practice this with a short piece first, because if you mess up you could lose your work.
As part of an exam, you may be asked how plants and animals create polymers from monomers.