Units of Measure
One of the next great advancements of human-kind came in the form of writing. There were others that were mastered first (like pottery, but to continue on our journey intelligently instead of trial and error the way our predecessors did, we need to take advantage of this great achievement. In order to replicate things as opposed to rubbing sticks or banging rocks, we need both numbers and units in addition to the words you are already taking advantage of by reading this page.
Credit for this section
|
1. Journal credit.
-You have evidence of detailed notes -50 points -You have a summary of key points (paragraph form) 25 points -Create your own problem for analysis, it can be real or fictional. You will be awarded points based on the number of conversions required. 1 step : 5 points 2 steps: 15 points 3 steps: 25 points |
2. Units Quiz
You will get 1 problem to solve by Unit Analysis. I will expect you to use both the conversions above AND equivalencies given to you in the problem. If you solve it, you pass, otherwise you fail. This quiz will be a 100 or a 0. You may retake quizzes once per week until they are passed or the grading period closes. Problems will not be the same. |
3. Units project
Ask your parents/uncle/cousin for the electricity bill or water bill, or find one online. Break down the entire bill by units and compare the costs of each piece. You should have an equivalence statement for each portion of the bill. For example, for every $1 that is paid on the water bill, how much is charged for sewer? Show the math and the equivalencies. This should be done as a pamphlet, poster, or similar. A copy of the bill is required. Grading: In addition to an equivalence for EVERY charge your project should have: 100: 5 conversions showing the cost per $1 of either electric or water 90: 4 conversions 80: 3 conversions 70: 2 conversions 50: 1 conversion |
What's it mean...?
Units are the basis for conveying all information about a measurement of any kind. And every measurement comes with 3 parts: the numerical part of the measurement, which is the number, the unit itself telling you what that number represents such as grams, liters, ounces, pounds, pH, etc., and a precision or degree of confidence. For example, if you are charged $4.87 for a meal at Jack in the Box, that is more precise than $5 because you are more confident in the measurement as shown by the number of decimal places. If you know how the units work and can find a conversion you should not only be able to skim through high school problems, but should also be able to work about 75% of the college problems you will encounter should you choose to go that route. If you learn nothing else in this course, this should be the thing you learn. Units are your friends, they tell you what is going on and how to change the problem to suit your needs. You could probably use another friend anyway, so there you go.
Common things you are expected to memorize... some of which you probably already know.
Since units are your friends, they are one of the rare things I will ask you to memorize in this course. I do this because you will need them when you exit high school--even if you never touch another science course again. You need them to do your taxes, compare interest rates for cars and houses, and a variety of other things in life. They tell you what is happening and how you can manipulate them to your advantage.
Each factor comes from a mathematical equality. For example 12 inches = 1 foot. So then (12 inches / 1 foot) must equal ONE since both the top and bottom of the fraction are equal. And we know that anything multiplied by 1 is itself, so this is how we get back and forth between units and convert them into something meaningful to what we are working with.
PREFIXES YOU SHOULD MEMORIZE:
prefix abbreviation factor scientific notation example
Kilo k 1000 10^3 there are 1000 meters in a kilometer
Centi c .01 or 1/100 10^-2 there are 100 centimeters in a meter
Milli m .001 or 1/1000 10^-3 there are 1000 milliliters in a liter
UNITS YOU SHOULD MEMORIZE
unit abbreviation what it measures measured with
gram g mass scale or balance
liter L volume graduated cylinder
meter m length/distance meter-stick or measuring tape
mole mol amount of chemical counting
second s time stop watch
Each factor comes from a mathematical equality. For example 12 inches = 1 foot. So then (12 inches / 1 foot) must equal ONE since both the top and bottom of the fraction are equal. And we know that anything multiplied by 1 is itself, so this is how we get back and forth between units and convert them into something meaningful to what we are working with.
PREFIXES YOU SHOULD MEMORIZE:
prefix abbreviation factor scientific notation example
Kilo k 1000 10^3 there are 1000 meters in a kilometer
Centi c .01 or 1/100 10^-2 there are 100 centimeters in a meter
Milli m .001 or 1/1000 10^-3 there are 1000 milliliters in a liter
UNITS YOU SHOULD MEMORIZE
unit abbreviation what it measures measured with
gram g mass scale or balance
liter L volume graduated cylinder
meter m length/distance meter-stick or measuring tape
mole mol amount of chemical counting
second s time stop watch
How to use them--analysis
1. Write the units of the answer you are looking for, followed by an equal sign. Be sure to leave room for numerical values.
2. Find an equivalence statement that matches what you have to what you want. This may require more than one to get there. For example if you wanted to turn inches into yards you would use inches to feet and then feet to yards.
3. Write the equivalence to the right of the equal sign, the unit you started with on the left should match the unit on the bottom of the right side so that they cancel when you go to do the algebra.
4. Keep adding equivalences until you get from the thing you started with to the thing you want.
5. Use your calculator to solve the numerical digits, use your pen to solve the units by cancelling. When you are done write down the whole number from the calculator and the final unit that is left over. If you have more than one unit left over that doesn't match what the problem calls for, you know you aren't done yet.
6. Round your numerical digits to the SAME NUMBER of digits as you started with in the problem.
EXAMPLES:
1. A kitchen recipe
Consider a large kitchen that requires 15 pounds of cheese to make 50 plates of food. Your equivalence becomes 15 pounds cheese / 50 plates food = 1. Since we are working at home and not the restaurant we only want to make 6 plates of food. How much cheese should we buy? We could choose several units for our answer such as ounces, grams, etc, but since cheese is sold by the 8oz bar or bag in the grocery store we should choose bars.
X bars cheese = 2 plates of food (15 pounds of cheese / 50 plates of food) (16 ounces cheese / 1 pound of cheese) ( 1bar of cheese / 8 ounces cheese) ** How to read the math** 1. When parenthesis are placed next to each other we multiply. 2. Whenever there is the "/" symbol we divide. 3. It is not necessary to do each step individually and all of them can be plugged into your calculator at once and the numerical values should look like this: 2*15/50*16/1*1/8 ... 2.4. Even thought the units don't go in the calculator they are still extremely important, if they don't cancel out (top and bottom) to be only the units you want then either something is missing or upside down and will get you an incorrect value, which means big money losses and massive food waste (or shortage).
= 2.4 bars of cheese ... so you need to buy 3 bars.
But wait Mister! Why do I have to round if I know exactly what I need, can't I just use the 2.4 bars of cheese? And in middle school I learned its 5 or more to raise the score... here its only 4, I should round down! Well yes that is true, until reality hits. The fact here is that we need to consider what we are doing before we round, and we're shopping...and the store won't sell you a part of a bar of cheese. And if you round down you won't have enough cheese...If you don't have enough cheese, your date won't be very impressed and you'll wind up single and alone filled with self-loathing, and we can't have that. So you'll actually have some cheese left over and make a quesadilla for tomorrow's lunch, which is awesome. Hooray for you! Your life was just improved through science!
CHEMISTRY EXAMPLE
How many grams of carbon are required to make 30g of cellulose? Any time we encounter an inequality of this kind we can find conversions for it. Later you will know where to find these but for now use the following conversions: 1 mole of carbon weighs 12 grams. 1 mole of cellulose weighs 30 grams. There is two moles of carbon for every one mole of cellulose. For this problem we don't even know what a mole is yet, but that shouldn't matter to you.
Start with the units of your answer:
1. g C
2. We know two things about Carbon. That 1 mol of it weighs 12 grams and that 1 mol of it equals 2 mols of cellulose. Since we are in grams, we need the conversion with grams.
3. X g C (1 mol C / 12 g C) .... this gets us out of grams and into moles of carbon.
4. We can now compare mols of carbon to something. Using grams would put us back where we started so that isn't useful and the only other thing we know is 2 mols of carbon make 1 mole of cellulose. we now have X g C (1 mol C / 12 g C) (1 mol cellulose / 2 mol C)
5. The next thing we have is 1 mol of cellulose for 30 grams of cellulose. X g C (1 mol C / 12 g C) (1 mol cellulose / 2 mol C) (30g cellulose / 1 mol cellulose) = 1.25 grams of carbon
=1.3 grams of Carbon (remember the rounding!)
You have actually just solved a common workplace level laboratory problem and:
1. You are still in high school
2. you don't even really know what a mole is yet
3. Most of these positions require a university degree
This is the power of units and analysis.
2. Find an equivalence statement that matches what you have to what you want. This may require more than one to get there. For example if you wanted to turn inches into yards you would use inches to feet and then feet to yards.
3. Write the equivalence to the right of the equal sign, the unit you started with on the left should match the unit on the bottom of the right side so that they cancel when you go to do the algebra.
4. Keep adding equivalences until you get from the thing you started with to the thing you want.
5. Use your calculator to solve the numerical digits, use your pen to solve the units by cancelling. When you are done write down the whole number from the calculator and the final unit that is left over. If you have more than one unit left over that doesn't match what the problem calls for, you know you aren't done yet.
6. Round your numerical digits to the SAME NUMBER of digits as you started with in the problem.
EXAMPLES:
1. A kitchen recipe
Consider a large kitchen that requires 15 pounds of cheese to make 50 plates of food. Your equivalence becomes 15 pounds cheese / 50 plates food = 1. Since we are working at home and not the restaurant we only want to make 6 plates of food. How much cheese should we buy? We could choose several units for our answer such as ounces, grams, etc, but since cheese is sold by the 8oz bar or bag in the grocery store we should choose bars.
X bars cheese = 2 plates of food (15 pounds of cheese / 50 plates of food) (16 ounces cheese / 1 pound of cheese) ( 1bar of cheese / 8 ounces cheese) ** How to read the math** 1. When parenthesis are placed next to each other we multiply. 2. Whenever there is the "/" symbol we divide. 3. It is not necessary to do each step individually and all of them can be plugged into your calculator at once and the numerical values should look like this: 2*15/50*16/1*1/8 ... 2.4. Even thought the units don't go in the calculator they are still extremely important, if they don't cancel out (top and bottom) to be only the units you want then either something is missing or upside down and will get you an incorrect value, which means big money losses and massive food waste (or shortage).
= 2.4 bars of cheese ... so you need to buy 3 bars.
But wait Mister! Why do I have to round if I know exactly what I need, can't I just use the 2.4 bars of cheese? And in middle school I learned its 5 or more to raise the score... here its only 4, I should round down! Well yes that is true, until reality hits. The fact here is that we need to consider what we are doing before we round, and we're shopping...and the store won't sell you a part of a bar of cheese. And if you round down you won't have enough cheese...If you don't have enough cheese, your date won't be very impressed and you'll wind up single and alone filled with self-loathing, and we can't have that. So you'll actually have some cheese left over and make a quesadilla for tomorrow's lunch, which is awesome. Hooray for you! Your life was just improved through science!
CHEMISTRY EXAMPLE
How many grams of carbon are required to make 30g of cellulose? Any time we encounter an inequality of this kind we can find conversions for it. Later you will know where to find these but for now use the following conversions: 1 mole of carbon weighs 12 grams. 1 mole of cellulose weighs 30 grams. There is two moles of carbon for every one mole of cellulose. For this problem we don't even know what a mole is yet, but that shouldn't matter to you.
Start with the units of your answer:
1. g C
2. We know two things about Carbon. That 1 mol of it weighs 12 grams and that 1 mol of it equals 2 mols of cellulose. Since we are in grams, we need the conversion with grams.
3. X g C (1 mol C / 12 g C) .... this gets us out of grams and into moles of carbon.
4. We can now compare mols of carbon to something. Using grams would put us back where we started so that isn't useful and the only other thing we know is 2 mols of carbon make 1 mole of cellulose. we now have X g C (1 mol C / 12 g C) (1 mol cellulose / 2 mol C)
5. The next thing we have is 1 mol of cellulose for 30 grams of cellulose. X g C (1 mol C / 12 g C) (1 mol cellulose / 2 mol C) (30g cellulose / 1 mol cellulose) = 1.25 grams of carbon
=1.3 grams of Carbon (remember the rounding!)
You have actually just solved a common workplace level laboratory problem and:
1. You are still in high school
2. you don't even really know what a mole is yet
3. Most of these positions require a university degree
This is the power of units and analysis.
|
Click the button to visit Texas A&M's department of chemistry review for solving problems with units.
|
Units and the periodic table
|
The periodic table is something you should be familiar with from middle school, and quite intimate with if you took IPC. Even if you remember neither of these sources, there is a great deal of information that can be gleaned from it by simply reading.
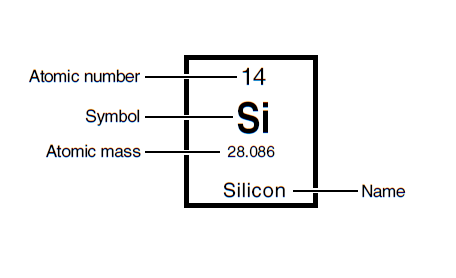
To the right, the unit we see is mass and from above this will be in grams. We can also get an equivalence statement from this, that is 28.086grams of Silicon = 1 unit of Silicon. For chemistry we count by 'moles' and we still don't what that is, nor do we really care at this point as we're still cave-people. Also for our course, rounding to whole numbers from the periodic table will typically be fine so we can use 28g Si = 1 mol Si. (remember in order to round you look at the next place, which is a zero here ... 28.0) Other information that is useful to us include the atomic number (recall this is how elements are identified and is equal to their number of protons), the symbol, and the name. As we have already encountered, it is recommended to look up names from symbols until you become comfortable with them as some names don't make much sense in English, such as Sodium symbol Na and Potassium symbol K. |
Quickly check yourself: if you have a sample of 3.4 moles of pure quartz, how much does it weigh?
1. Recall from STONE that quartz is SiO2 and that the silicates are the most common thing found in the earth's crust 2. Count the atoms you have present, Si =1 and O =2 3. Look up their values on the periodic table, 1 mol Si =28g and 1 mol O = 16g 4. Add up the value for 1 mol SiO2 : 28g+16g+16g= 60g (remember the little subscript 2 beneath the O means that there are 2 Oxygens present in the compound) 5. Convert: if 1 mol SiO2 = 60g then 3.4 mol x 60g/1mol = 204g SiO2 6. Round to the number of digits you started with: 200g SiO2 |
PRACTICE PROBLEMS
You can find a variety of practice problems with solutions online by searching for either "unit factor analysis" or "dimensional analysis", but I will also give you a few here.
1. Imagine that you are driving your car in Canada or Mexico. As you're driving along, you notice that the speed limit signs have numbers like 120 (on the highway) and 50 (in the city). As you start to speed up, you realize that the signs are in km/hour. Unfortunately, your speedometer only reads in mi/hour, how fast are you allowed to go in mi/hr on the highway? Use 1km = 0.6214mi.
2. An empty tank measures 20 cm x 50 cm at the base. If 1000 liters of water are added to the tank, how deep will it be in meters? 1mL = 1cm^3
3. The football field is 100 yards long and 160 feet wide. If a half inch of rain falls on the field, how many gallons of water have to drain from the field? There are 231 in^3 in one gallon.
4. You are a nurse and have a 140lb patient that requires a dopamine infusion at a rate of 5 mg per pound of body weight per every 15 minutes. If the IV bag contains 800 grams of dopamine in a 500mL solution, at what infusion rate, in mL/Hour should the pump be set?
ANSWER
1. about 75mph
2. 10 meters
3. 14,900 gallons
4. 1.75 mL/Hour
You can find a variety of practice problems with solutions online by searching for either "unit factor analysis" or "dimensional analysis", but I will also give you a few here.
1. Imagine that you are driving your car in Canada or Mexico. As you're driving along, you notice that the speed limit signs have numbers like 120 (on the highway) and 50 (in the city). As you start to speed up, you realize that the signs are in km/hour. Unfortunately, your speedometer only reads in mi/hour, how fast are you allowed to go in mi/hr on the highway? Use 1km = 0.6214mi.
2. An empty tank measures 20 cm x 50 cm at the base. If 1000 liters of water are added to the tank, how deep will it be in meters? 1mL = 1cm^3
3. The football field is 100 yards long and 160 feet wide. If a half inch of rain falls on the field, how many gallons of water have to drain from the field? There are 231 in^3 in one gallon.
4. You are a nurse and have a 140lb patient that requires a dopamine infusion at a rate of 5 mg per pound of body weight per every 15 minutes. If the IV bag contains 800 grams of dopamine in a 500mL solution, at what infusion rate, in mL/Hour should the pump be set?
ANSWER
1. about 75mph
2. 10 meters
3. 14,900 gallons
4. 1.75 mL/Hour