|
Notes to the right: you may ignore sections not outlined in the objective (the bottom two)
|
OBJECTIVE:
Predict molecular structure for molecules with linear, bent, trigonal planar, trigonal pyramidal or tetrahedral electron pair geometries using Valence Shell Electron Pair Repulsion (VSEPR) theory.
Extra credit:
Make a video OR POSTER to show the different structures using your body to represent attached atoms and electron pairs. Save your video to your google drive and then SHARE IT with [email protected], include your class period and group member's names!!
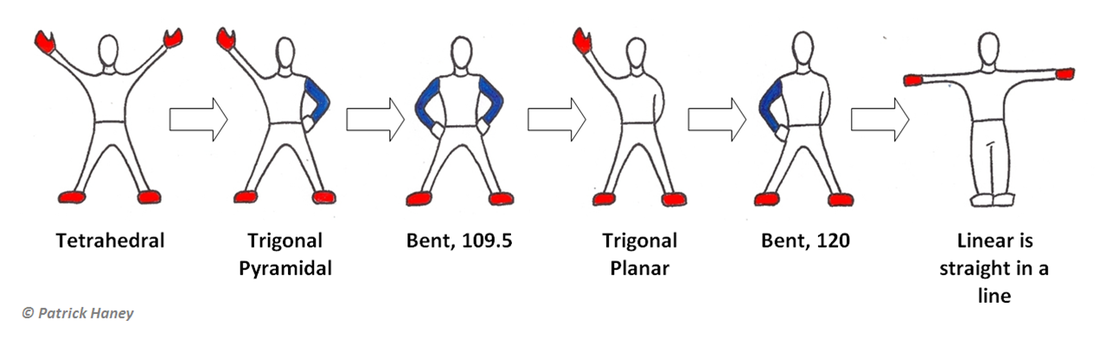
In the image above the red portions represent elements attached to the central atom, while the blue portions represent unbonded pairs of electrons on the Lewis Diagram.
Predict molecular structure for molecules with linear, bent, trigonal planar, trigonal pyramidal or tetrahedral electron pair geometries using Valence Shell Electron Pair Repulsion (VSEPR) theory.
Extra credit:
Make a video OR POSTER to show the different structures using your body to represent attached atoms and electron pairs. Save your video to your google drive and then SHARE IT with [email protected], include your class period and group member's names!!
In the image above the red portions represent elements attached to the central atom, while the blue portions represent unbonded pairs of electrons on the Lewis Diagram.
