OBJECTIVES:
1. Explain Mendeleeve's use of chemical and physical properties in the development of the Periodic Table
2. Use the Periodic Table to identify and explain the properties of chemical families, including alkali metals, alkaline earth metals, halogens, noble gases, and transition metals.
3. Use the Periodic Table to identify and explain periodic trends, including atomic and ionic radii, electronegativity, and ionization energy.
1. Explain Mendeleeve's use of chemical and physical properties in the development of the Periodic Table
2. Use the Periodic Table to identify and explain the properties of chemical families, including alkali metals, alkaline earth metals, halogens, noble gases, and transition metals.
3. Use the Periodic Table to identify and explain periodic trends, including atomic and ionic radii, electronegativity, and ionization energy.
|
Compare the elements and their respective groups. Use your seated chemical families to help you. Why are these elements in the same groups?
Use the buttons below to guide you, what properties do they have in common? For the above link, ensure you click on group numbers, blocks, and period numbers. Also click metal/nonmetal
|
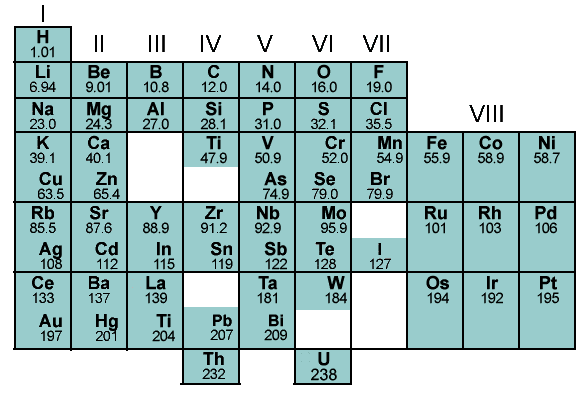
Dmitri Mendeleev created this first table of the elements. Notice the holes... why did he place gaps there?
View the video below... why are the alkali metals grouped together? What common property do they have? |