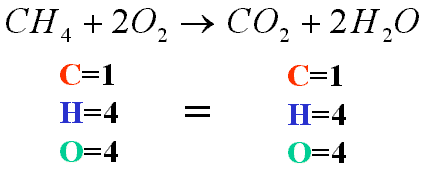Objectives:
1. Name ionic compounds
2. Name covalent compounds
3. Name polyatomic compounds
1. Name ionic compounds
2. Name covalent compounds
3. Name polyatomic compounds
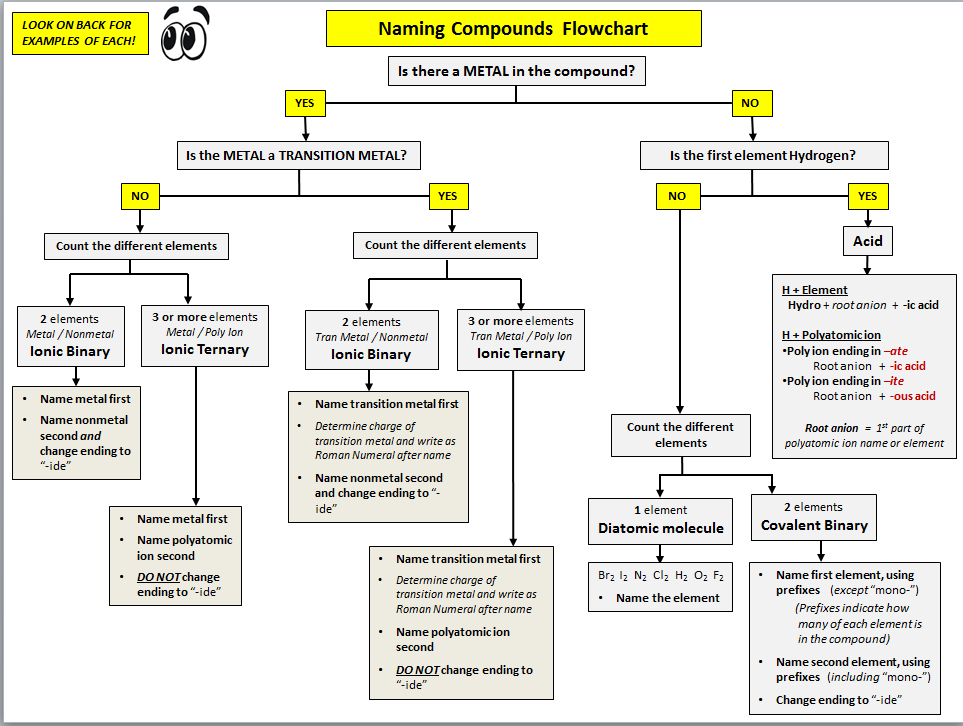
REMEMBER THE FIRST STEP IS ALWAYS IDENTIFY THE TYPE OF COMPOUND: ionic, covalent, or polyatomic?
Formulas come from STABLE Lewis diagrams... don't forget how they workIONIC: metal is present and electrons transfer
COVALENT: only nonmetals and electrons are shared |
NamingFollow the flow chart to name your compounds. Click below for a zoomable copy of the flow chart
|
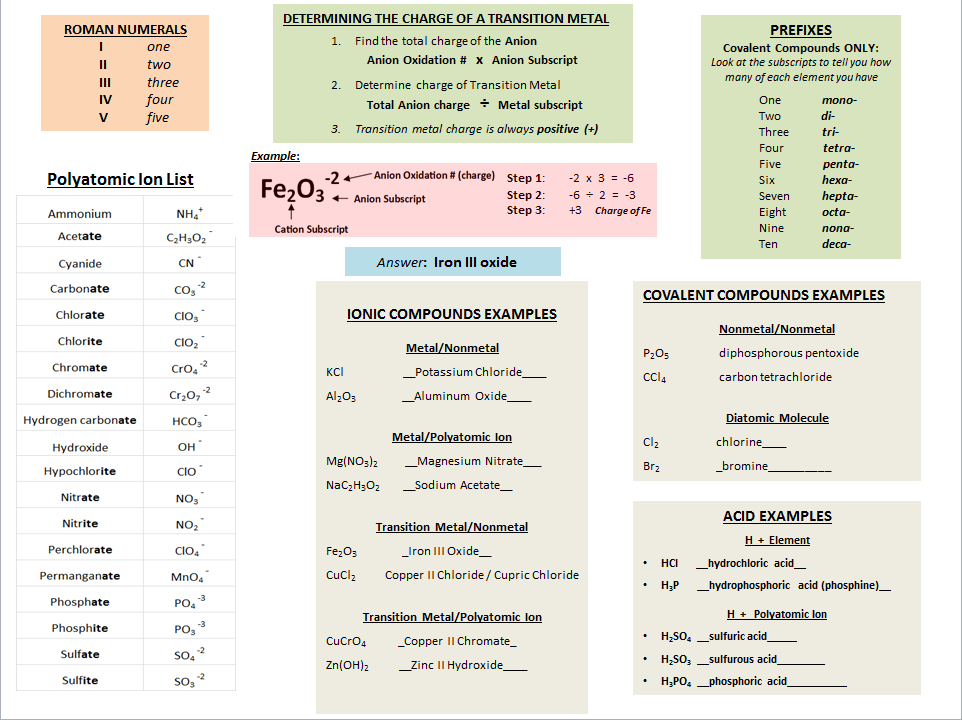
ReferencesDon't forget your reference material!! Highlight the polyatomic section on the back of your formula chart, Roman numerals and prefixes you need to KNOW.
|